Bo, Y., Zhang, H., Li, Y., Reva, Y., Xie, L., Guldi, D.M.
Angewandte Chemie, 136(13), e202313936
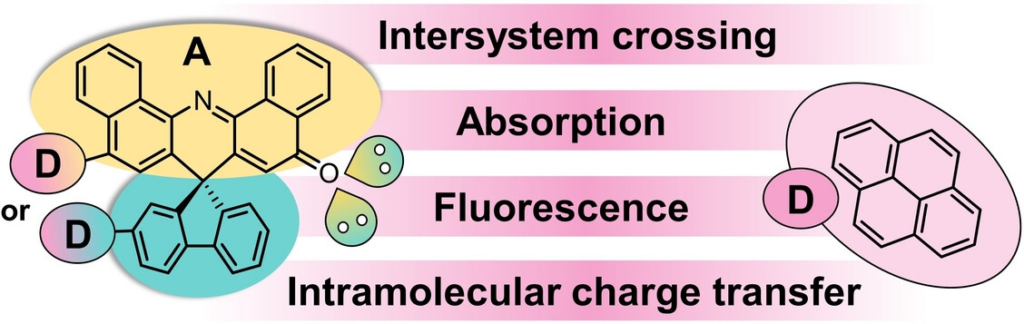
In this work, we prepared a series of electron donor-acceptor systems based on spiro[fluorene-9,7’-dibenzo[c,h]acridine]-5’-one (SFDBAO). Our SFDBAOs consist of orthogonally positioned fluorenes and aromatic ketones. By fine-tuning the substitution of electron-donating pyrenes, the complex interplay among different excited-state decay channels and the overall impact of solvents on these decay channels were uncovered. Placing pyrene, for example, at the aromatic ketones resulted in a profound solvatochromism in the form of a bright charge-transfer (CT) emission spanning from yellow to red-NIR. In contrast, a dark non-emissive CT was noted upon pyrene substitution at the fluorenes. In apolar solvents, efficient triplet-excited state generation was observed for all SFDBAOs. Either charge transfer was concluded to mediate the intersystem crossing (ISC) in the case of pyrene substitution or the El-Sayed rule was applicable when lacking pyrene substitution as in the case of SFABAO. In polar solvents, charge separation is the sole decay upon pyrene substitution. Moreover, competition between ISC and CT lowered the triplet-excited state generation in SFDBAO
Bo, Y., Zhang, H., Li, Y., Reva, Y., Xie, L., Guldi, D.M.
Angewandte Chemie, 136(13), e202313936
In this work, we prepared a series of electron donor-acceptor systems based on spiro[fluorene-9,7’-dibenzo[c,h]acridine]-5’-one (SFDBAO). Our SFDBAOs consist of orthogonally positioned fluorenes and aromatic ketones. By fine-tuning the substitution of electron-donating pyrenes, the complex interplay among different excited-state decay channels and the overall impact of solvents on these decay channels were uncovered. Placing pyrene, for example, at the aromatic ketones resulted in a profound solvatochromism in the form of a bright charge-transfer (CT) emission spanning from yellow to red-NIR. In contrast, a dark non-emissive CT was noted upon pyrene substitution at the fluorenes. In apolar solvents, efficient triplet-excited state generation was observed for all SFDBAOs. Either charge transfer was concluded to mediate the intersystem crossing (ISC) in the case of pyrene substitution or the El-Sayed rule was applicable when lacking pyrene substitution as in the case of SFABAO. In polar solvents, charge separation is the sole decay upon pyrene substitution. Moreover, competition between ISC and CT lowered the triplet-excited state generation in SFDBAO